Stem Cell Therapy for Hip Pain: What Patients Should Know
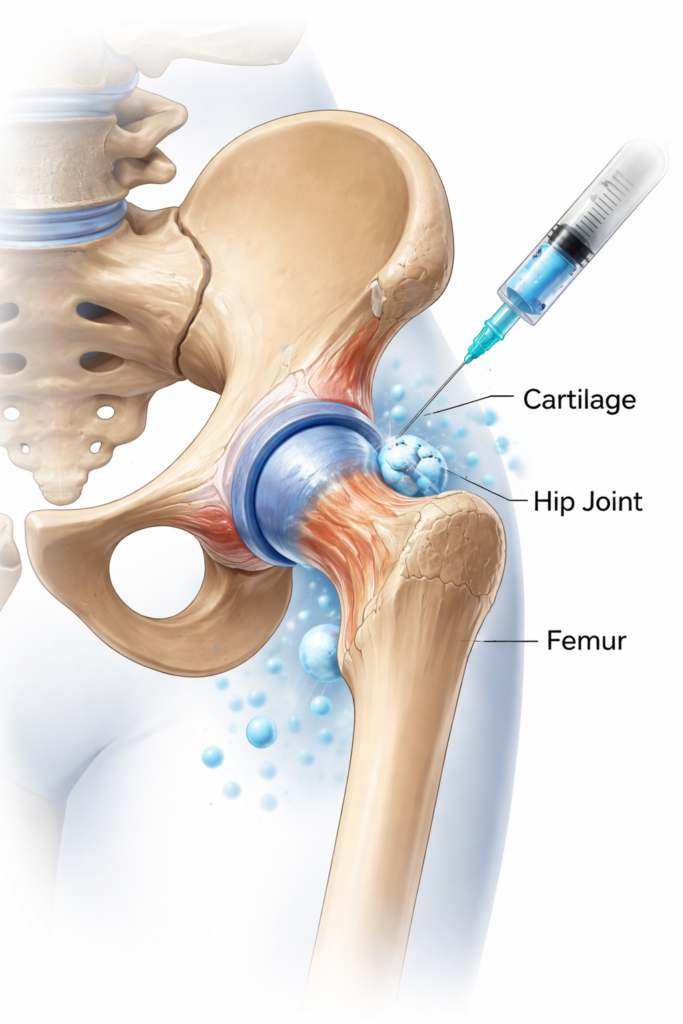
Chronic hip pain — whether related to osteoarthritis, cartilage degeneration, or post-injury discomfort — can limit mobility and interfere with daily activities. Stem cell therapy for hip pain is an investigational regenerative medicine approach being studied as a potential non-surgical option to support hip joint environments affected by degeneration or inflammation.
Chronic hip pain can limit mobility, interfere with daily activities, and significantly affect quality of life. For individuals who have not found adequate relief from conservative treatments, stem cell therapy has emerged as an area of regenerative medicine being actively studied for hip-related conditions.
For a broader national overview, patients may also review our page on stem cell therapy in Mexico, which outlines regulatory context, safety considerations, and general evaluation processes.
What Is Stem Cell Therapy for Hip Pain?
Stem cell therapy is a regenerative medicine approach being researched for its potential role in supporting the body’s natural repair and signaling processes. In hip-related applications, stem cell–based procedures typically involve administering stem cells into or around the hip joint under medical supervision.
- Participate in cellular communication
- Influence inflammatory pathways
- Support joint environments affected by degeneration or injury
Hip Conditions Patients Commonly Ask About
Patients who inquire about stem cell therapy for hip pain often report underlying hip conditions such as:
Hip osteoarthritis
A degenerative joint condition that may lead to stiffness, reduced range of motion, and persistent discomfort, particularly during weight-bearing activities.
Cartilage degeneration
Gradual wear or damage to hip cartilage that can affect joint cushioning and contribute to pain with movement.
Labral tears
Injuries to the cartilage rim of the hip joint that may cause clicking sensations, instability, or pain with certain motions.
Chronic joint inflammation
Ongoing inflammatory processes within the hip joint that may contribute to discomfort and functional limitations over time.
Post-injury or overuse-related hip pain
Hip pain that develops after trauma or repetitive stress, sometimes persisting despite rest or conservative care.
Degenerative joint conditions such as osteoarthritis may affect multiple joints. Some patients also explore whether stem cell therapy has been studied for stem cell therapy for knee arthritis.
What the Procedure Involves
While specific protocols may vary, stem cell therapy for hip pain typically follows a structured clinical process designed to support patient safety and informed decision-making.
The procedure commonly involves:
- Medical screening and review of prior imaging such as X-rays or MRI
- Evaluation by licensed medical professionals to assess hip joint condition
- Preparation of stem cells from ethically sourced tissue
- Administration under clinical protocols, sometimes using imaging guidance
- Post-procedure monitoring and follow-up recommendations
An important consideration for many patients is how stem cells are sourced. You can learn more about ethical sourcing and donor considerations in our article, Where Do Stem Cells Come From?
Who May Be a Candidate for Stem Cell Therapy?
A patient may be considered for evaluation if they:
- Experience chronic hip pain despite conservative care
- Are exploring non-surgical options
- Are not ideal candidates for hip replacement
- Understand the investigational nature of stem cell therapy
Risks, Limitations, and Important Considerations
Stem cell therapy involves potential risks and limitations, including:
- Infection
- Inflammatory response
- No improvement in symptoms
- Variable outcomes
Patients should understand that:
- Stem cell therapy is not FDA-approved for hip pain
- Research is ongoing
- Results reported by others may not be typical
Additional considerations include the lack of standardized treatment protocols, variation based on the severity of the underlying hip condition, and differences in individual biological response. Some patients may not experience noticeable symptom changes, and response timelines can vary widely, sometimes taking several weeks or months. Regulatory status is an important consideration. Patients can review current information on whether stem cell therapy is FDA approved.
What Research Says About Stem Cell Therapy for Hip Pain
Research into stem cell therapy for hip osteoarthritis and related joint conditions is ongoing. Some studies have reported promising trends in symptom support and joint function, but results are not consistent across all patients. At this time, stem cell therapy for hip pain remains investigational and is not FDA-approved.
Why Some Patients Explore Stem Cell Therapy in Mexico
Some patients from the United States consider treatment outside the U.S. due to:
- Regulatory differences
- Access to investigational therapies
- Cost considerations
- Availability of specialized regenerative clinics
Rejuvacell Medical Group provides care to international patients with an emphasis on:
- Medical screening
- Patient education
- Safety protocols
Medical Oversight and Patient Care
All procedures at Rejuvacell Medical Group are:
- Reviewed by licensed medical professionals
- Conducted under clinical protocols
- Evaluated individually
Frequently Asked Questions
Is stem cell therapy for hip pain FDA approved?
Stem cell therapy is not FDA-approved for hip pain. Many procedures remain investigational.
Can stem cell therapy replace hip replacement?
Some patients explore regenerative options before surgery, but surgery may still be necessary depending on severity.
How soon after treatment do patients typically resume normal activities?
Recovery experiences vary depending on the individual and the nature of the procedure. Some patients resume normal activities within a short period, while others may require additional time based on medical guidance.
How long does it take to see changes after treatment?
Clinical response timelines vary widely. Some patients may notice changes over several weeks or months, while others may not experience noticeable improvement.
Is imaging used during the evaluation process?
Imaging studies such as X-rays or MRI are commonly reviewed during the medical evaluation to better understand hip joint structure and overall condition.
Request a Hip Pain Consultation
If you are experiencing chronic hip pain and want to learn more about stem cell therapy, you may request a confidential consultation with a medical coordinator.
- No obligation
- Patient-focused discussion
- Medical review required
