Stem Cell Therapy for Knee Pain: Benefits, Risks, and What to Expect
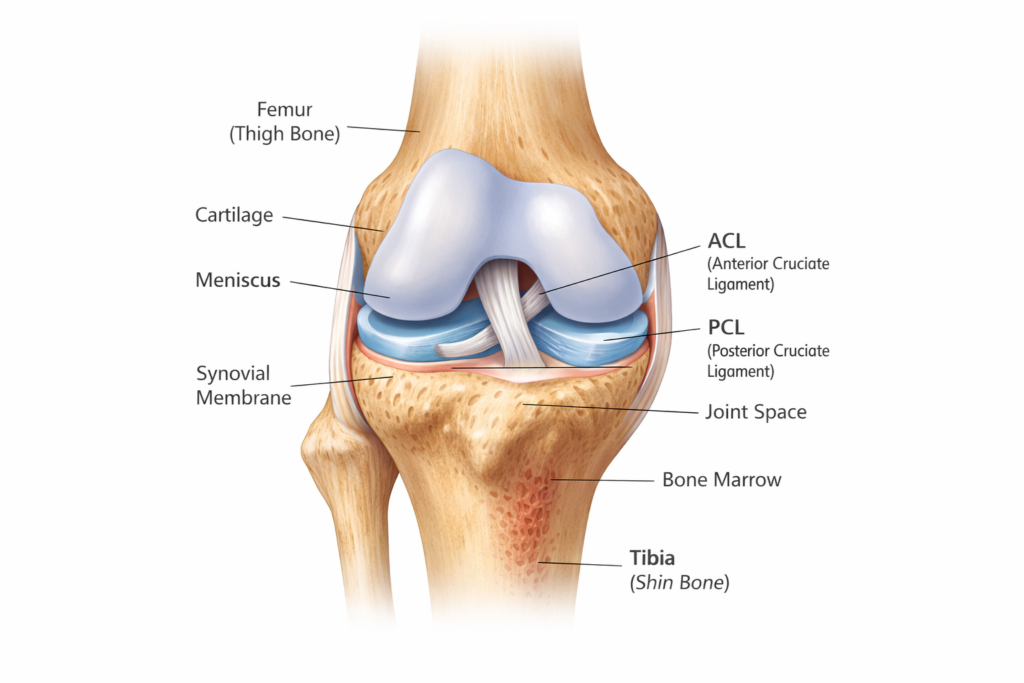
Chronic knee pain can make everyday activities—walking, climbing stairs, or standing for extended periods—feel increasingly difficult. For many patients, conventional treatments such as medications, physical therapy, or injections may not provide lasting relief.
Stem cell therapy for knee pain is an investigational approach within regenerative medicine that is being studied for its potential to support the body’s natural repair processes. While it is not a guaranteed solution, some patients explore this option when looking for alternatives to surgery or other non-surgical approaches for joint conditions.
Patients dealing with joint degeneration often explore how these therapies apply to broader conditions such as arthritis: Stem Cell Therapy for Arthritis
Understanding Stem Cell Therapy for Knee Pain
Stem cell therapy is a developing area of regenerative medicine focused on how certain cells may help support tissue environments affected by injury or degeneration.
In the context of knee pain, stem cell–based procedures are being studied for their potential role in:
- Supporting cellular signaling
- Modulating inflammation
- Contributing to tissue repair environments
For an overview of how stem cell therapies are currently classified in the United States: FDA stem cell overview
How Stem Cell Therapy Works in the Knee
Stem cell procedures for knee pain generally follow a structured clinical process:
- Initial Evaluation & Imaging:
A licensed provider reviews medical history, performs a physical exam, and often orders imaging (X-ray or MRI) to assess joint condition and guide planning. - Stem Cell Harvesting:
Stem cells are collected from ethically sourced tissue (e.g., bone marrow or adipose); this is done in a clinical setting. - Processing & Preparation:
The collected tissue is processed in a controlled lab to concentrate stem cells and prepare them for targeted administration. - Guided Injection into the Knee:
Using imaging guidance such as ultrasound, the provider injects the prepared cells into the specific area of the knee requiring attention. - Post-Procedure Monitoring & Follow-Up:
Patients are observed briefly, given after-care instructions (e.g., rest, icing), and scheduled for follow-up to monitor recovery and outcomes.
In general, stem cell–based procedures typically involve:
- Harvesting stem cells from ethically sourced tissue
- Processing them in a controlled laboratory setting
- Administering them into the affected knee joint
At the same time, research is ongoing into how stem cells may:
- Support cellular signaling
- Contribute to tissue repair environments
- Interact with inflammation pathways
Degenerative joint conditions, such as knee arthritis, are also being studied in regenerative medicine research.
Does Stem Cell Therapy for Knee Pain Actually Work?
Current research on stem cell therapy for knee pain shows mixed but evolving results.
Some studies suggest that certain patients may experience:
- Reduced pain levels
- Improved joint function
- Better mobility over time
However:
- Evidence remains limited and variable
- Results differ significantly between individuals
- No standardized treatment protocols currently exist
Patients should also understand the regulatory status of these therapies before making a decision: Is Stem Cell Therapy FDA Approved?
For ongoing clinical research and published studies: PubMed
What Stem Cell Therapy Can and Cannot Do
Stem cell therapy is often discussed as a regenerative solution, but understanding its realistic role is essential.
What it may help with:
- Supporting reduction of inflammation
- Improving joint comfort in some patients
- Enhancing mobility over time
What it may not do:
- It does not reliably regenerate cartilage in advanced conditions
- It does not cure osteoarthritis
- It does not guarantee symptom improvement
Patients may also explore how similar approaches are used in other joint conditions: Stem Cell Therapy for Shoulder Pain
Conditions Commonly Evaluated
Knee Osteoarthritis and Chronic Knee Pain
Patients who inquire about stem cell therapy for knee pain often have conditions such as:
Knee osteoarthritis
A degenerative joint condition that may cause ongoing knee pain, stiffness, and reduced mobility, particularly during weight-bearing activities. Some patients explore stem cell therapy for arthritis as part of ongoing research into regenerative approaches.
Cartilage degeneration
Gradual wearing of the cartilage that cushions the knee joint, which can lead to discomfort, swelling, and limited range of motion over time.
Meniscus-related knee pain
Pain associated with damage or wear to the knee’s meniscus, often experienced during twisting movements, squatting, or prolonged activity.
Chronic joint inflammation
Persistent inflammation in the knee joint that may result in pain, stiffness, and swelling, sometimes worsening after physical activity.
Post-injury knee discomfort
Ongoing knee symptoms that remain after a previous injury, such as strain or trauma, even once the initial injury has healed.
Who May Be a Candidate — and Who May Not Be
Patients who may be evaluated include:
- Individuals with chronic knee pain not responding to conservative care
- Patients seeking to delay or avoid surgery
- Those with mild to moderate joint degeneration
Patients who may not be ideal candidates include:
- Individuals with advanced joint damage
- Patients expecting guaranteed results
- Those with conditions that may affect healing
What Results Can Patients Expect?
Patient experiences vary widely.
Some individuals report:
- Gradual reduction in pain
- Improved joint mobility
- Better tolerance for daily activities
However:
- Some patients experience minimal or no improvement
- Effects may take weeks or months
- Results are not guaranteed
Recovery Timeline After Treatment
Recovery timelines vary, but general patterns include:
First few days:
- Mild swelling or discomfort
- Activity modification
First few weeks:
- Gradual return to light activity
Several months:
- Evaluation of response and symptom changes
Following medical guidance is important for optimal recovery.
Risks, Limitations, and Safety Considerations
However, while stem cell–based therapies are an area of ongoing research, it is important to understand their current limitations and potential risks.
-
Lack of standardized protocols:
Treatment approaches may vary between providers, as standardized clinical protocols for stem cell–based knee procedures are still being developed. -
Variation based on condition severity:
Individual responses can differ depending on factors such as the severity of the knee condition, overall joint health, and patient-specific medical factors. -
Uncertain and variable timelines:
Some patients may not experience notable symptom change, and response timelines vary widely, with potential effects—if any—sometimes taking weeks or months. -
Possible risks and side effects:
As with any medical procedure, potential risks may include infection, inflammatory response, or lack of symptom improvement.
For official safety information: FDA
Why Patients Consider Stem Cell Therapy in Mexico
Some U.S. patients explore treatment options outside the United States due to:
- Regulatory differences
- Access to investigational therapies
- Cost considerations
- Availability of specialized regenerative clinics
Rejuvacell Medical Group treats international patients while emphasizing:
- Patient education
- Medical screening
- Safety protocols
Medical Oversight & Experience
All procedures are:
- Reviewed by licensed medical professionals
- Conducted under clinical protocols
- Evaluated on a case-by-case basis
Stem Cell Therapy vs Other Treatment Options
Patients often compare stem cell therapy with other approaches depending on their condition.
Understanding how knee arthritis is evaluated and treated can help guide decision-making: Can Stem Cell Therapy Help Knee Arthritis?
What the Patient Journey Looks Like
The process typically includes:
- Initial consultation
- Medical and imaging review
- Eligibility assessment
- Discussion of treatment options
- Follow-up and monitoring
Frequently Asked Questions
Is stem cell therapy for knee pain FDA approved?
Stem cell therapy is regulated differently depending on the country. Many stem cell procedures remain investigational and are not FDA-approved for knee pain.
How long does the procedure take?
Treatment protocols vary and are discussed during consultation, depending on individual evaluation.
Will I avoid knee surgery?
Some patients explore stem cell therapy as an alternative, but outcomes vary and surgery may still be necessary.
How long does it take to see changes after treatment?
Response timelines vary widely depending on the individual, the severity of the knee condition, and other medical factors. Some patients may notice changes within several weeks, while others may take months. Not all patients experience noticeable symptom changes.
Is imaging, such as an MRI, used during the consultation process?
In many cases, imaging studies such as X-rays or MRI scans are reviewed as part of the consultation process. Imaging can help providers better understand joint condition and determine whether a patient may be a candidate for further evaluation.
Request a Knee Pain Consultation
If you are experiencing chronic knee pain and want to learn more about stem cell therapy, you can request a confidential consultation with a medical coordinator.
- No obligation
- Patient-focused discussion
- Medical review required
