Chronic Tendonitis Stem Cell Therapy: What Patients Should Know
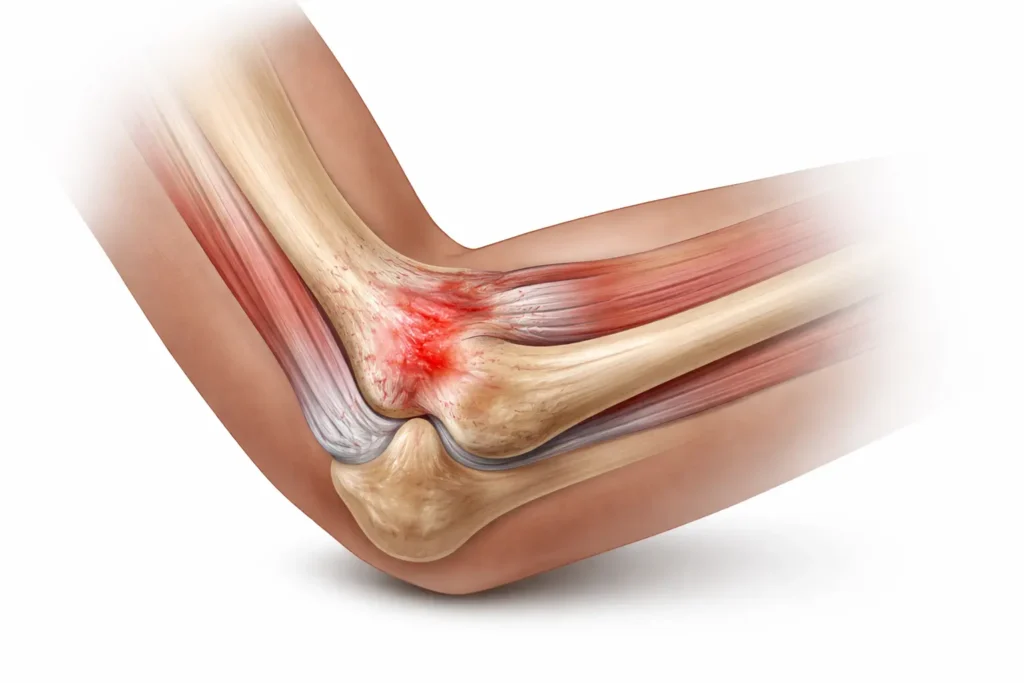
Chronic tendonitis can cause persistent pain, stiffness, and reduced function that interfere with work, sports, and daily activities. Researchers are studying chronic tendonitis stem cell therapy as an investigational, non-surgical option to support tendon tissue affected by degeneration or poor healing.
For a broader national overview, patients may also review our page on stem cell therapy in Mexico, which outlines regulatory context, safety considerations, and general evaluation processes.
What Is Stem Cell Therapy for Chronic Tendonitis?
Researchers currently study stem cell therapy for chronic tendonitis as part of regenerative medicine research focused on supporting the body’s natural repair processes. Some patients with persistent tendon pain explore chronic tendonitis stem cell therapy as an investigational, non-surgical option when conservative treatments—such as rest, physical therapy, or injections—have not provided lasting relief.
Importantly, researchers continue to evaluate stem cell therapy, and outcomes vary. It is not a guaranteed treatment or cure for chronic tendonitis.
For this reason, providers encourage patients to understand the investigational nature of stem cell–based procedures and to undergo a medical evaluation.
How Stem Cells Are Studied for Chronic Tendonitis
Current studies examine how stem cells interact with damaged tendon tissue and inflammatory pathways involved in long-standing tendon pain. At the same time, researchers continue to study how these biological processes vary depending on tendon location, injury severity, and individual patient factors.
What the Procedure Involves:
General Steps in Stem Cell Therapy for Chronic Tendonitis
- Initial Evaluation & Imaging
A licensed provider reviews medical history, performs a focused physical examination, and may order imaging such as MRI or diagnostic ultrasound to evaluate tendon structure, degeneration, and surrounding soft tissue involvement. - Stem Cell Harvesting
Medical providers collect stem cells from ethically sourced tissue, such as bone marrow or adipose tissue, in a controlled clinical environment following established medical protocols. - Processing & Preparation
Laboratory specialists process the harvested tissue in a regulated setting to concentrate stem cells and prepare them for targeted administration to the affected tendon area. - Image-Guided Injection into the Tendon
Using ultrasound guidance, the prepared stem cells are precisely injected into or around the degenerated tendon tissue to support accurate placement and procedural safety. - Post-Procedure Monitoring & Follow-Up
After the procedure, medical staff monitor patients briefly, provide after-care instructions, and may schedule follow-up visits to assess symptom progression and tendon function over time.
In general, these procedures involve providers harvesting stem cells from ethically sourced tissue, processing them in a controlled laboratory setting, and administering them to affected tendon structures.
At the same time, research is ongoing into how stem cells may:
- Support cellular signaling
- Contribute to tendon repair environments
- Interact with inflammatory pathways involved in chronic tendon pain
Tendon Conditions Studied in Stem Cell Research
In many cases, patients who inquire about chronic tendonitis stem cell therapy experience persistent tendon pain related to degeneration, overuse, or prior injury. The following tendon conditions are commonly discussed in regenerative medicine research. Each condition is different, and eligibility depends on individual medical factors.
Chronic Tendon Degeneration and Tendinosis
Tendinosis refers to degenerative changes within the tendon structure that may cause ongoing pain, stiffness, and reduced strength. These changes often develop over time due to repetitive stress, inadequate healing, or age-related tissue degeneration.
In some cases, tendon degeneration affects the shoulder region, particularly in rotator cuff–related injuries. Some patients also explore stem cell therapy for shoulder pain when tendon damage contributes to ongoing shoulder symptoms.
Chronic Tendonitis and Overuse Injuries
Long-standing tendon inflammation or micro-damage caused by repetitive activity may lead to recurring pain and functional limitation. Overuse injuries are common in both occupational and athletic settings.
Post-Injury Tendon Pain
Ongoing tendon symptoms may persist after a previous strain, tear, or traumatic injury, even once the initial injury has healed. In these cases, degenerative tissue changes may contribute to chronic discomfort.
Long-standing tendon inflammation or micro-damage caused by repetitive activity may lead to recurring pain and functional limitation. In some patients, these tendon changes may coexist with nearby joint degeneration, prompting them to also research stem cell therapy for arthritis as part of a broader musculoskeletal evaluation.
Post-Surgical Tendon Symptoms
Some patients experience lingering tendon pain or reduced mobility following tendon-related surgical procedures. Regenerative approaches are sometimes explored as supportive care during recovery.
Who May Be a Candidate for Stem Cell Therapy?
Stem cell therapy for chronic tendonitis is not appropriate for everyone. A medical evaluation is required to determine whether this investigational approach may be suitable based on individual health factors, tendon condition, and treatment history.
A patient may be considered for evaluation if they:
- Have chronic tendon pain not responding to conservative care
- Experience symptoms lasting three months or longer
- Are seeking non-surgical options
- Wish to delay or avoid tendon-related surgery
- Understand the investigational nature of stem cell therapy
Risks and Limitations to Consider
While researchers continue to study stem cell–based therapies, patients should understand their current limitations and potential risks. Because of this, individual responses vary, timelines remain uncertain and outcomes cannot be guaranteed.
-
Lack of standardized protocols:
Treatment approaches may vary between providers, as standardized clinical protocols for stem cell–based tendon procedures are still being developed. -
Variation based on tendon severity:
Response may differ depending on factors such as the extent of tendon degeneration, tissue quality, injury chronicity, and overall musculoskeletal health. -
Uncertain and variable timelines:
Some patients may not experience noticeable symptom changes, and response timelines—if any—can vary from weeks to months. -
Possible risks and side effects:
As with any medical procedure, potential risks may include infection, inflammatory response, discomfort at the injection site, or lack of symptom improvement.
Why Patients Consider Stem Cell Therapy in Mexico
Some U.S. patients explore stem cell therapy options outside the United States due to differences in regulatory frameworks, access to investigational regenerative therapies, and cost considerations. Availability of specialized regenerative medicine clinics is also a factor for some individuals.
When seeking care abroad, patients should prioritize education, medical screening, and provider experience as part of the decision-making process.
Rejuvacell Medical Group treats international patients while emphasizing:
- Patient education
- Medical screening
- Safety protocols
Medical Oversight & Experience
Licensed medical professionals evaluate all stem cell–based procedures within a structured clinical framework to support patient safety and appropriate medical decision-making.
All procedures are:
- Reviewed by licensed medical professionals
- Conducted under clinical protocols
- Evaluated on a case-by-case basis
Frequently Asked Questions
Is stem cell therapy for chronic tendonitis FDA approved?
Stem cell therapy for chronic tendonitis is not FDA approved as a standard treatment. It is considered an investigational approach that continues to be studied in regenerative medicine research. Regulatory status may vary depending on how the cells are processed and used, and patients should review these considerations with a qualified medical provider.
How long does the procedure take?
Stem cell–based procedures for chronic tendon conditions are typically performed on an outpatient basis and generally take about 60 to 90 minutes, including preparation, image-guided injection, and brief post-procedure observation.
Can stem cell therapy help avoid tendon surgery?
Some patients with chronic tendon degeneration or overuse injuries explore stem cell therapy as a non-surgical option. While certain individuals may delay or avoid surgery, results vary, and stem cell therapy cannot guarantee that surgery will be avoided. Severe tendon tears or advanced degeneration may still require surgical intervention.
How long does it take to notice changes after treatment?
Response timelines vary between patients. Some individuals report changes within several weeks, while others may take several months to notice differences, if any. Because stem cell therapy is investigational, outcomes and timelines cannot be predicted or guaranteed.
Is imaging used during the evaluation and procedure?
Yes. Imaging such as MRI or diagnostic ultrasound is commonly used during the evaluation process to assess tendon structure and degeneration. Ultrasound guidance is also frequently used during the injection to support accurate placement.
What the Patient Journey Typically Looks Like
Typically, many patients begin with an initial consultation and medical screening to review symptoms, medical history, and imaging when appropriate. During this process, a medical provider evaluates tendon condition, prior treatments, and overall health factors.
If eligibility is determined, a personalized treatment approach may be discussed based on individual needs, clinical findings, and investigational protocols.
Request a Tendonitis Consultation
If you are experiencing chronic tendon pain and want to learn more about stem cell therapy, you may request a confidential consultation with a medical coordinator.
- No obligation
- Patient-focused discussion
- Medical review required
